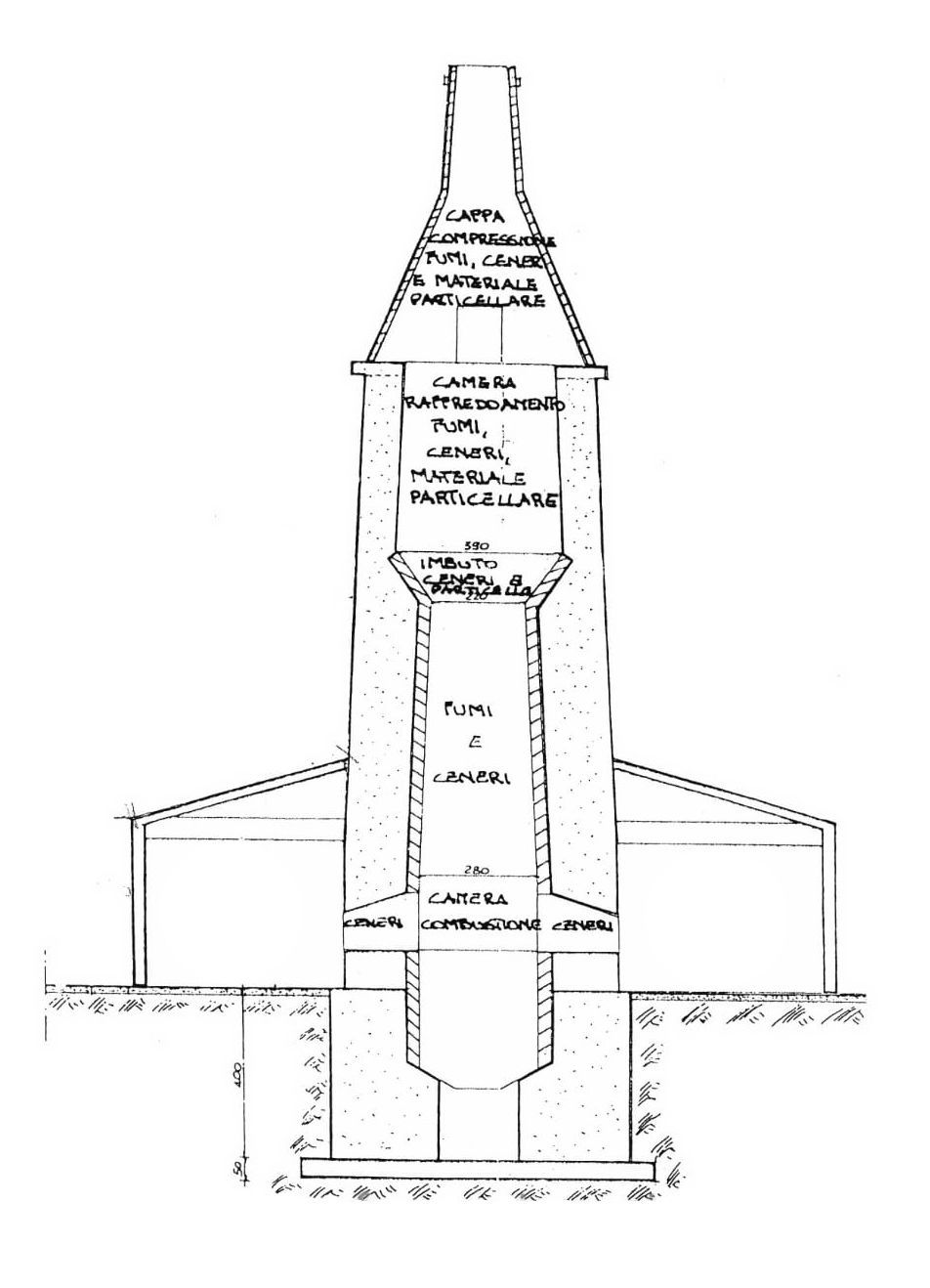
THE COOKING OF THE CALCAREOUS STONES CRYSTALS (CALCIUM CARBONATE) BEGINS WITH INSERTION IN THE RAISED PART OF THE OVEN (FUNNEL), for the first important phase of limestone drying, remaining for a long time and with low temperatures ranging from 70 ° / 120 ° Approximately ° C, after descending they enter the upper part of the cooking chamber, where they remain for a long time for the second drying phase with rising temperatures ranging from approximately 140 ° / 250 °C.
THIS METHOD OF SOFT DRYING LIMESTONE IT IS CARRIED OUT FOR SEVERAL DAYS, used for millennia, it is feasible only with these ancient vat ovens with a continuous cooking cycle with natural draft, it serves to prepare and avoid limestone in the subsequent phases of increasing temperatures from sudden thermal shocks, abruptly igniting the limestone stones and causing them to fast water loss.
ALSO OUR ANCIENT TIN OVEN with its system for separating the combustion chamber from the cooking chamber, it avoids putting the flames in direct contact with the material to be cooked, "this contact," stone flame "would compromise the quality and purity of the limestone, as well as the whiteness, even for rising ashes.
THIS ANCIENT PHASE OF LIMESCALE PREPARATION at the end of cooking, it will deliver a clean, porous, crumbly, reactive precious lime oxide, with single grains, very reactive in the hydration phase.
WHILE IN MODERN FORCED VENTILATION OVENS, FUELED BY GAS OR OTHER FUEL, the calcium oxide that is obtained is cooked at high temperatures ranging from 1,150 ° C to 1,300 ° C (CALCE BAKED TO DEATH),
IN ADDITION, THE STONE INTRODUCED IS SMALL, that, coming into contact with the aspirated flame and mechanically directed into the inside of the cooking chamber of the calcareous material, which by overheating excessively causes the rapid loss of water, the calcium oxide obtained from it, will have, the more it is high cooking, greater quantities of compact and adherent masses of large, not porous grains and with less capacity for reactivity in hydration in the quenching phase.
AFTER DRYING, THE LIME IS READY TO ENTER THE INTERMEDIATE PART of the cooking chamber with temperatures ranging from about 300 / 400C °, it remains in this area for the necessary period, then by gravity it reaches the central part of the cooking chamber reaching the optimum temperature around 500/600 ° C, at this temperature the calcium carbonate begins to gradually dissociate into calcium oxide and release carbon dioxide.
AT THE END THEY REACH THE END PART OF THE OVEN FOR FINAL COOKING, PROGRESSIVELY REACHING THE OPTIMAL TEMPERATURE, APPROXIMATELY 700 / 860C °. This cooking phase varies from 10 to 12 hours, to complete the transformation of calcium carbonate CaCO3 into calcium oxide CaO, this reaction takes place by thermal decomposition of the limestone (endothermic reaction) producing a porous homogeneous reactive calcium oxide CaO releasing carbon dioxide CO2
In the reaction CaCO3 -> CaO + CO2.
FOR THE PURITY OF OUR CALCIUM OXIDE (CAO)> 95.1%,
WE OBTAIN AN EXCELLENT TRADITIONAL CALCIUM FATTY LIME.
AT THE END OF COOKING THE VIVE CAO PLATES ARE REDUCED FROM THEIR INITIAL WEIGHT BY 40%, it also causes the loss of carbon C and oxygen O atoms, and the living clods take on a very porous crumbly consistency. Furthermore the gray color variations of the limestone are lost and the stones become mostly white.
THE COMPLETE COOKING CYCLE TAKES PLACE IN 14/15 DAYS,
ACCORDING TO EXTERNAL CLIMATIC CONDITIONS
presence of wind energy, a climatic factor that accentuates the chimney effect of the cooking system of the natural draft oven.
ONCE OBTAINED AN EXCELLENT HOMOGENEOUS BASIC CALCIUM OXIDE
TRADITIONAL, IT IS IMMERSED IN ABUNDANT WATER,
and due to the strong basic reactive reaction (natural exothermic) it brings the living clods to the boil, releasing a violent heat and the disintegration of the cooked stones (living clods) into pulp due to the expansive effect of the transformation from CaO oxide to calcium hydroxide Ca (OH )2
In the reaction CaO + H2O -> Ca (OH) 2.
IN THIS SHUTDOWN PHASE,
LIME OXIDE dissolves in slaked lime, hydroxide of lime, Ca (OH) 2, with the formation of single porous macro - prismatic crystals (portlandite).
The obtained CA (OH) 2 DENSE CALCE MILK is immersed in the maturation tanks, for the months necessary for its use.
CRYSTALS OF CALCE HYDROXIDE CA (OH) 2 DURING THE MATURATION PHASE (MINIMUM PER MONTHS 3) undergo important morphological and scaling changes with the formation of single porous tubular / hexagonal micro - crystals (portlandite).
WITH THIS ANCIENT METHOD YOU OBTAIN A TRADITIONAL LIME GREASE CREAM, which is dense, full-bodied, homogeneous, thixotropic in its natural creaminess and almost greasy softness, excellent in the viscosity of sealing of the aggregates and with excellent adhesion to the masonry, improving in plasticity, workability and water retention.
THE CARBONATION OF THE CREAM OF GRASSELLO FAT DI CALCE SETTING AND HARDENING REACTION of calcium hydroxide, takes place in the following reaction thus composed from the formal point of view.
Ca (OH) 2 + CO2 ---> CaCO3 + H2O, RETURNING TO THE INITIAL STATE OF CALCIUM CARBONATE CACO3 with the loss of H2O water.
TRADITIONAL BINDER
Natural, Ecological, Porous, Breathable, Insulating, Dehumidifying,
Antibacterial, Antifreeze.
ONCE APPLIED IT DOESN'T SHAPE caustic soluble salts neither efflorescence nor water retention.